
Elevated temperatures were finally applied to cure the thermoset. Solvents such as ethanol were used to dissolve TA in the resin, or TA was mixed in the resin at elevated temperatures to obtain a good homogeneous mixture. Most of the studies that used TA as a curing agent were confronted with the issue of the low compatibility of TA in the resin. However, only moderate thermal and mechanical properties were obtained such as a maximum T g of 77 ☌ and a storage modulus of 1103 MPa at 25 ☌. cured ESO with tannic acid to obtain fully bio-based EP. The use of TA to cure EVO has also been researched. found for the DGEBA cured with TA thermoset a T g >200 ☌, which is within the range of several commercially available high T g EPs, such as dicyandiamide and 4,4'-diaminodiphenyl sulfone. TA as an alternative curing agent for the commercially available DGEBA has also been investigated. used commercial TA as a curing agent to prepare EP with glycerol polyglycidylether (GPE) and sorbitol polyglycidylether (SPE). The high potential of TA as a bio-based substitute for petrochemical-based and toxic curing agents has already been highlighted, and several studies have been carried out to generate epoxy thermosets from TA. TA has the advantage of being already industrially isolated and an inexpensive chemical. TA has potentially 25 galloyl hydroxyl groups capable of cross-linking, and the rigid gallic structure suggests the formation of highly crosslinked networks and strong mechanical properties. TA is a nontoxic polyphenolic compound found in nuts, galls, seeds, and tree bark, and is affirmed as generally recognized as safe (GRAS). Ī particular focus is given in this publication to the bio-sourced tannic acid (TA). Furthermore, the T g of the epoxy cured with a bio-based hardener is often too low for high-performance application. Biologically sourced and nontoxic hardeners used industrially are quite rare as their synthesis and isolation mechanisms are complex and constrain their commercialization. Finding a bio-based, nontoxic, and non-harmful hardener capable of creating rigid networks with ELO to use bio-based thermosets in high-performance applications is of high interest. A hardener with a rigid structure such as an aromatic ring structure is, therefore, more suited than a hardener with a long aliphatic chain to obtain a stiff and highly crosslinked network with ELO. Due to the flexible structure of the triglycerides in the epoxidized linseed oil, a hardener with a rigid chemical structure is needed to create a thermoset that exhibits high stiffness and strength. ELO is the most suitable EVO for high-performance applications due to its high amount of epoxy groups in its linolenic acid chain. Generally, the mechanical and thermal properties of EPs depend strongly on their chemical structures, which are determined by the types of epoxy resins and curing agents used. Their long aliphatic chains impart flexibility, and the location of their oxirane rings in non-terminal positions is responsible for their lower reactivity than conventional DGEBA epoxy resins, which limit their practical use in structural applications. As a result, there is an increasing demand for bio-based epoxy thermoset alternatives.Įpoxidized vegetable oils (EVO) such as epoxidized soybean oil (ESO), epoxidized canola oil, or epoxidized linseed oil (ELO) have attracted great attention as an epoxy resin replacement due to their bio-based, unharmful nature, high availability, and relatively low price. Additionally, toxicity in cured epoxy materials may not be avoided as incomplete consumption of curing agents may remain. Moreover, some commonly used curing agents such as polyamine, polyamide, and anhydride hardeners are toxic before curing. However, considerable research has been and is still being focused on using bio-based compounds to replace BPA, which is classified as an endocrine disruptor. Glycerol, a byproduct from the biodiesel industry, can be used to produce ECH. The raw materials for DGEBA, bisphenol A (BPA), and epichlorohydrin (ECH) are petroleum-based and cause adverse effects on living organisms and ecosystems. 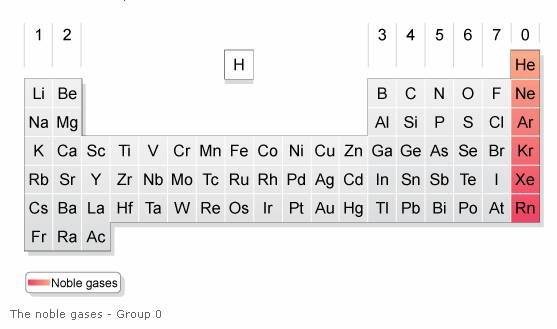
Ībout 90% of the epoxy thermoset is derived from diglycidyl ether of bisphenol A (DGEBA). Its market is expected to witness further growth in the forecast period of 2021–2026, growing at a Compound Annual Growth rate (CAGR) of 5%, and is projected to reach ≈4.3 million metric tons by 2026. EPs represent a prominent place in the thermosetting market. 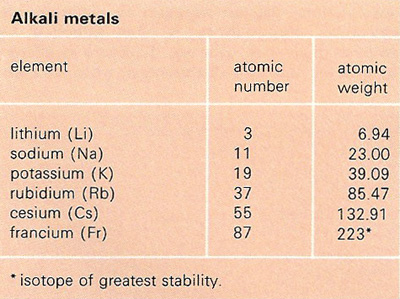
Epoxy thermosets (EP) are well established in a large variety of applications, including electronic encapsulation, paints, coatings, adhesives, sealants, and composite materials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
